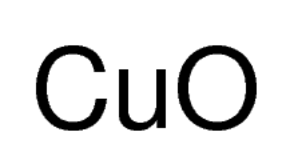![Copper-coated steel electrodes from copper(II) oxide and [Hbet] [NTf 2... | Download Scientific Diagram Copper-coated steel electrodes from copper(II) oxide and [Hbet] [NTf 2... | Download Scientific Diagram](https://www.researchgate.net/publication/347290413/figure/fig9/AS:993688419323914@1613925189507/Copper-coated-steel-electrodes-from-copperII-oxide-and-Hbet-NTf-2-a-in-sulfolane.png)
Copper-coated steel electrodes from copper(II) oxide and [Hbet] [NTf 2... | Download Scientific Diagram

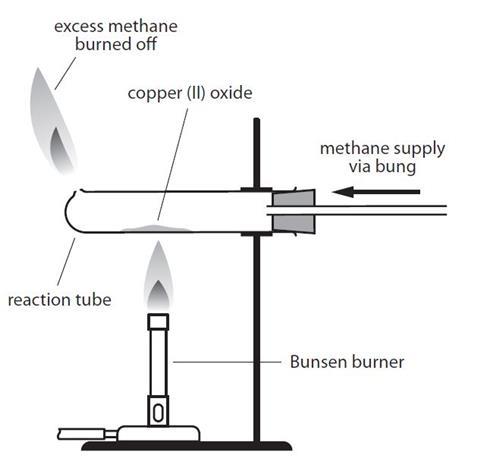
Copper(II) oxide reacts with ammonia to give copper, water and nitrogen. CuO((s)) +NH(3(g)) to xCu((s))+yH(2)O((l)) +zN(2(g)) The value of (x + y + z) is

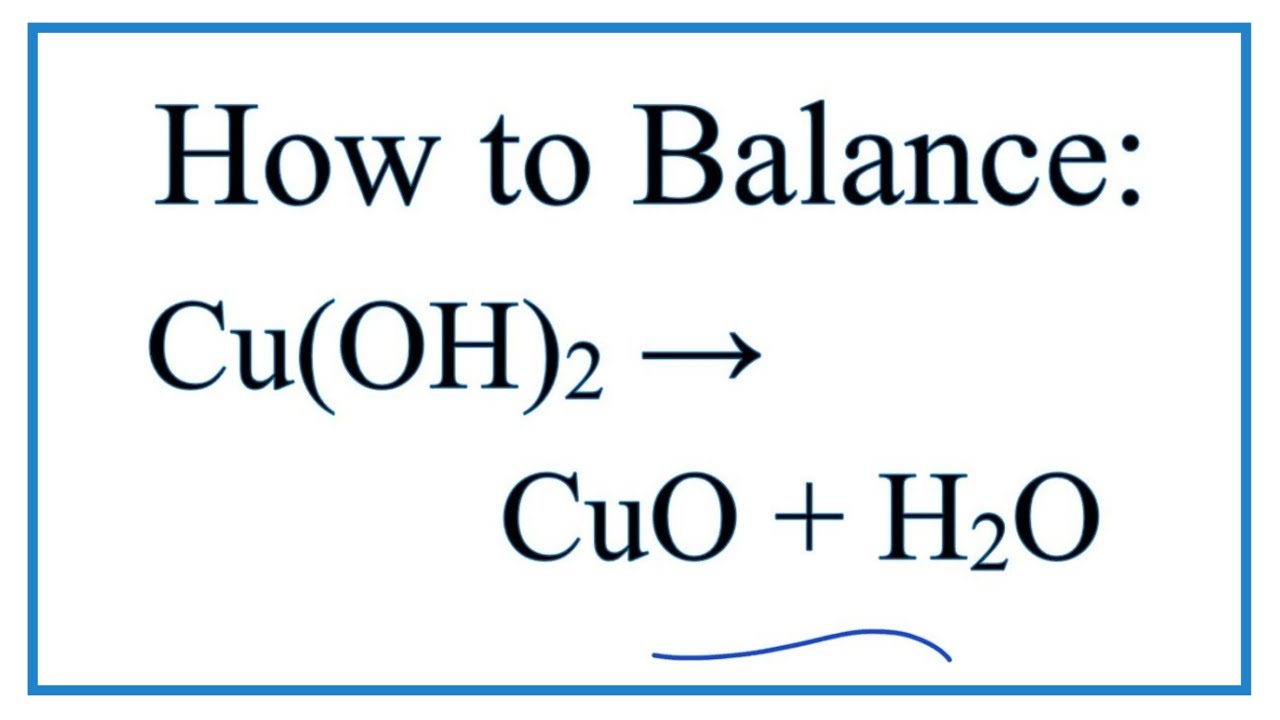
SOLVED: Using the 3 equations given in the experimental section for the process starting with copper (II) sulfate pentahydrate and ending with copper (II) oxide a5 = model, write equations for the
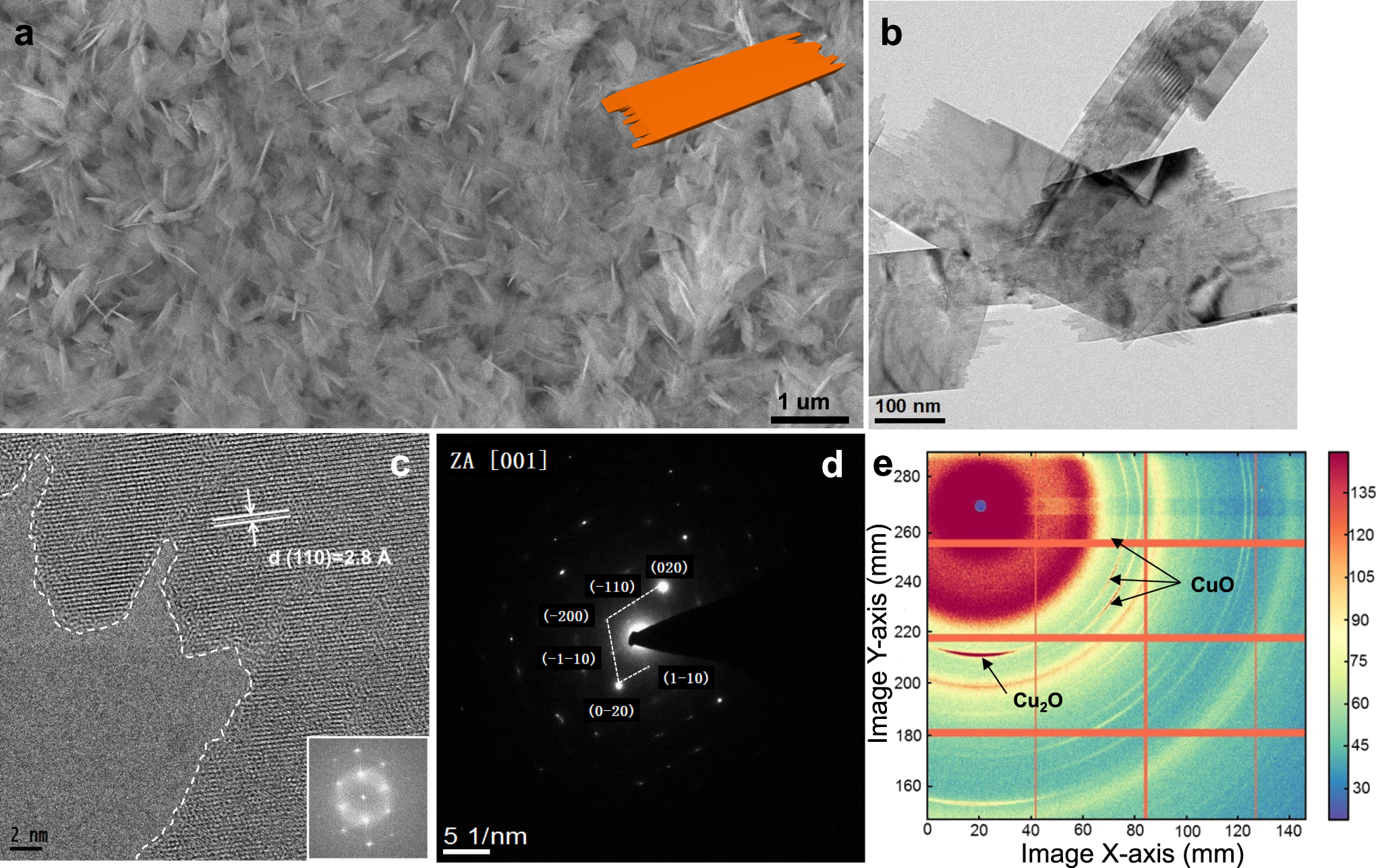
Morphology and mechanism of highly selective Cu(II) oxide nanosheet catalysts for carbon dioxide electroreduction | Nature Communications